Effective 5PL Easily Computes Asymmetric Cell-Based Potency Assay Curves
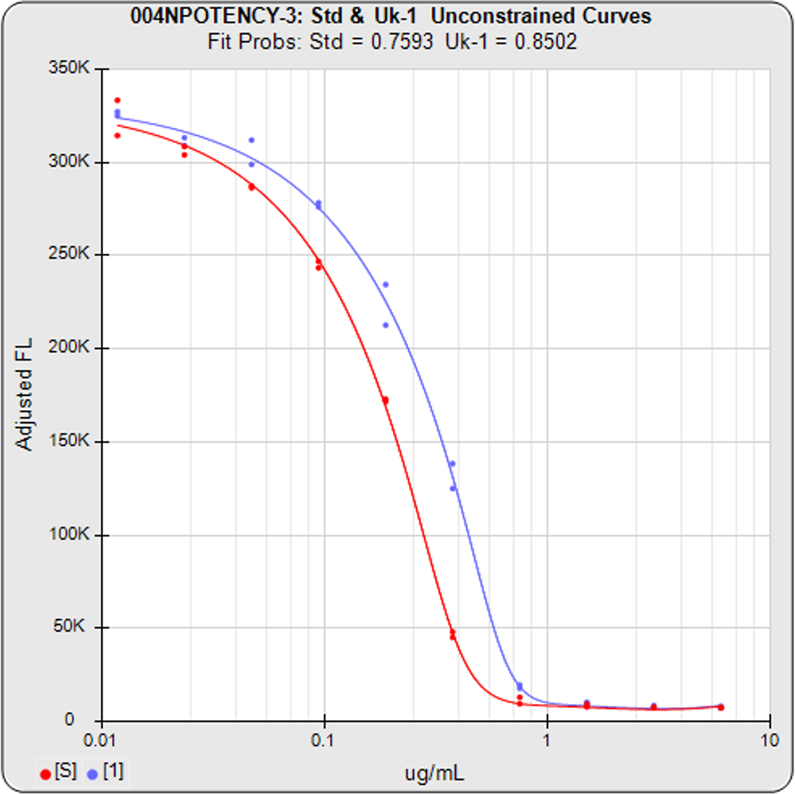
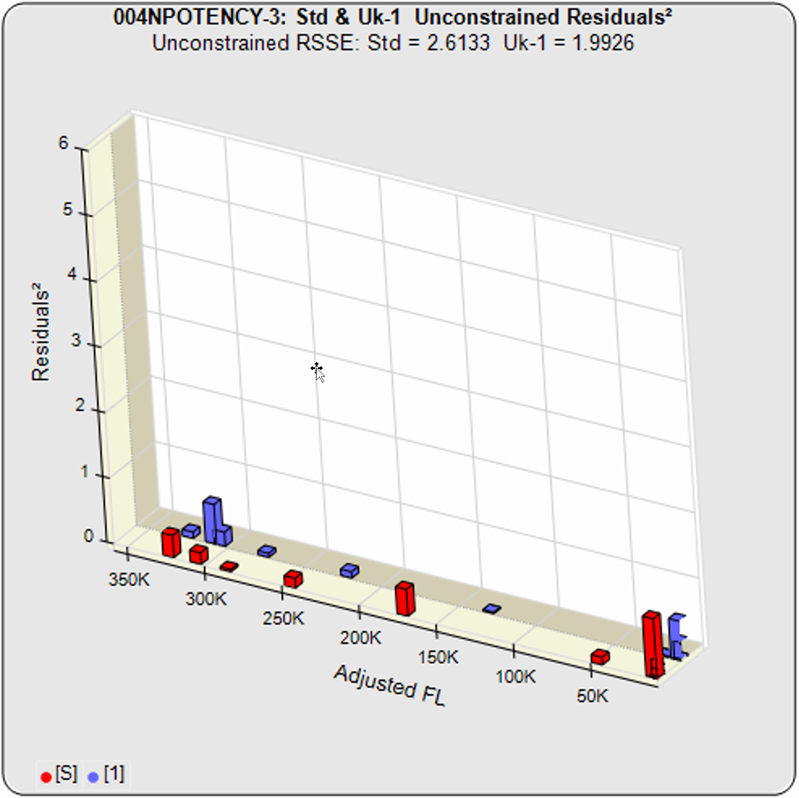
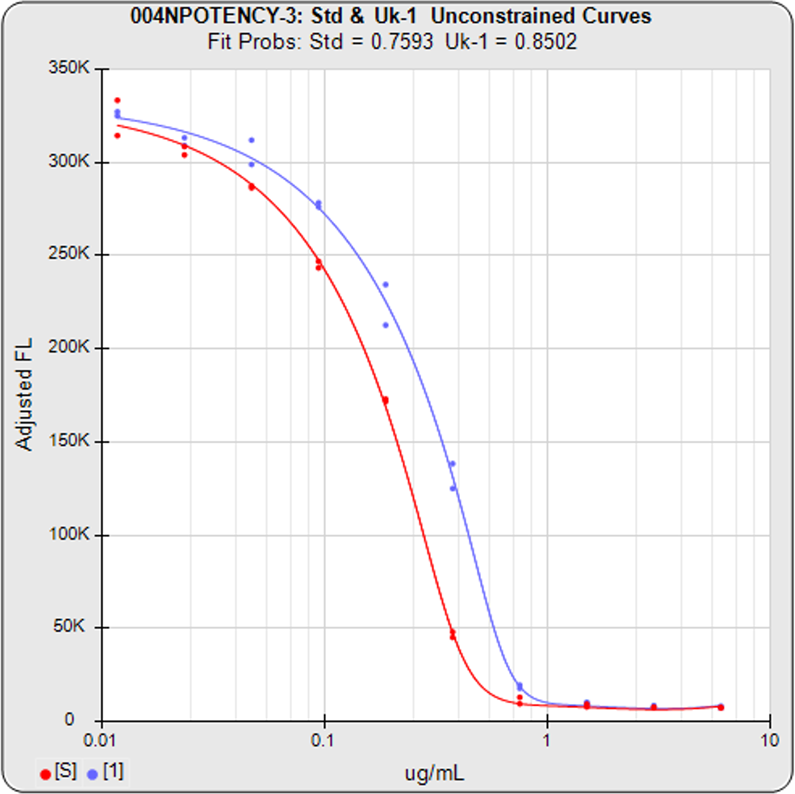
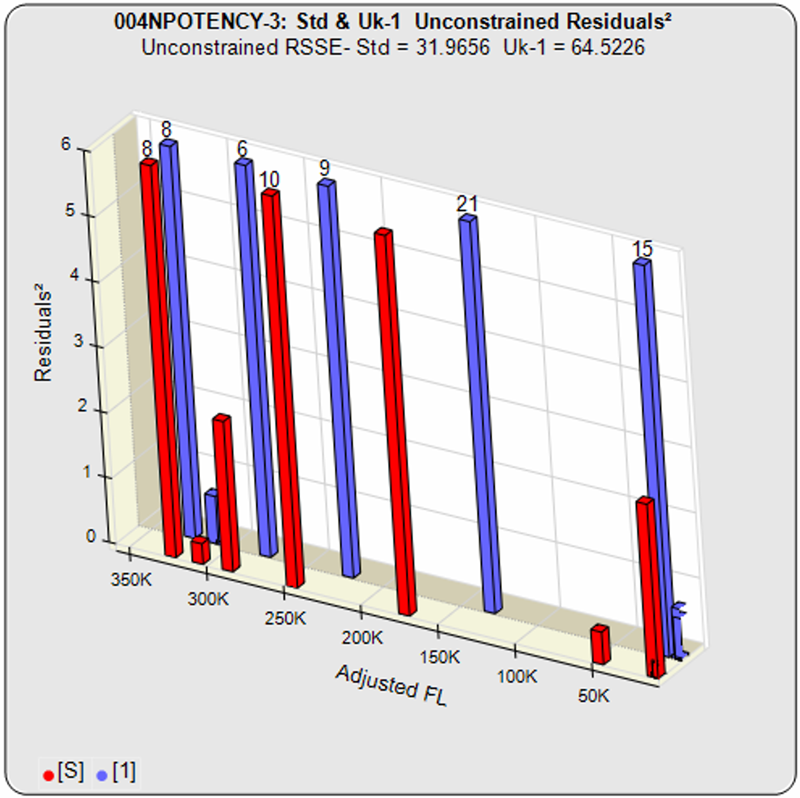
Some cell-based bioassays are characterized by a very sharp transition at the upper or lower asymptote and a more gradual transition to the other asymptote, as shown in the curves above. The squared residual errors of each dilution (red and blue bars) show the significant difference a 5PL can make fitting this asymmetric data. The fifth parameter in a 5 parameter logistic curve enables the curve to fit two distinctly different end shapes and yield a reliable determination of similarity. The 4 parameter logistic curve forces both ends to have the same shape, resulting in a bad fit even for the middle points.
It is good to have options. And having the option to use a 5PL with asymmetric potency assay curves can avoid difficulties and save time. STATLIA MATRIX 5.0 includes three methods referenced by the USP 1032, 1033, 1034 and EP 5.3 guidelines for computing the parallelism between curves:
- Equivalence Method (4PL)
- Chi-Square Method (5PL or 4PL)
- F Test (5PL or 4PL)
For the Equivalence Method, STATLIA MATRIX uses Monte Carlo simulations to compute more accurate confidence intervals, in addition to a robust 4PL.
For the Chi-Square Method, STATLIA MATRIX uses appropriate weighting and an effective 5PL or 4PL to provide a direct measure of the amount of similarity between curves and a reliable metric for parallelism.
For the F Test, STATLIA MATRIX uses Finney’s model.
No matter which method you use with STATLIA MATRIX, you are always getting the gold standard for 5PL and 4PL logistic models and the most accurate and reliable computations and analysis for your potency assays.
See manuscript on the software’s weighted 5PL model that has been requested more than 5,000 times and cited more than 300 times: The Five Parameter Logistic: A Characterization And Comparison With The Four Parameter Logistic.